Eli Lilly Surges 7% as Mounjaro and Zepbound Sales Smash Q1 Expectations
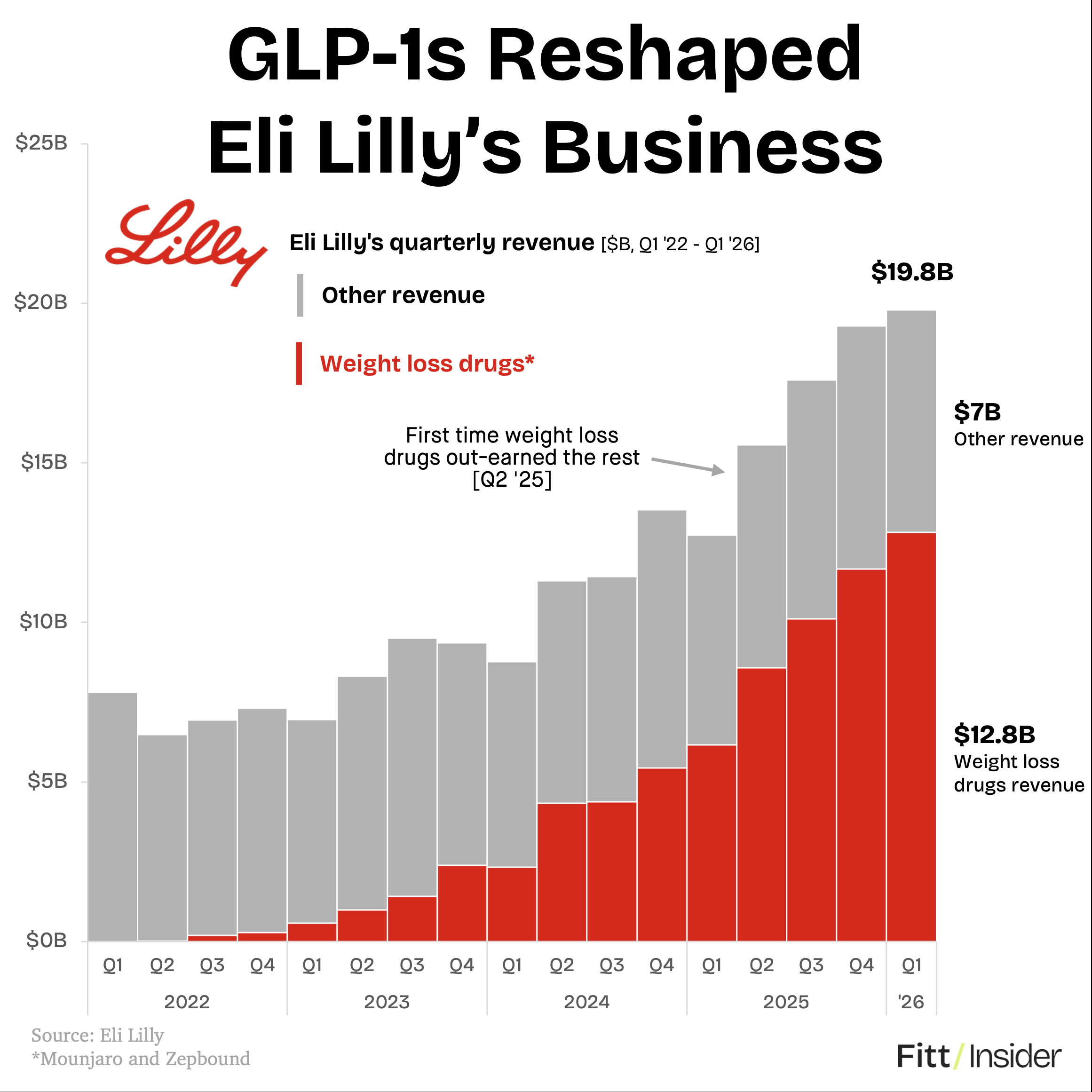
Eli Lilly shares surged nearly 7% on May 1 after the Indianapolis-based pharmaceutical company reported first-quarter 2026 earnings that demolished Wall Street forecasts, with its blockbuster diabetes drug Mounjaro posting 125% year-over-year revenue growth and its obesity treatment Zepbound delivering 80% growth -- results that cement Lilly's position as the dominant force in the GLP-1 drug market and raise fresh questions about how long competitors can remain relevant.
Background
The GLP-1 receptor agonist drug class -- which includes Mounjaro and Zepbound from Eli Lilly and Ozempic and Wegovy from Danish rival Novo Nordisk -- has transformed the pharmaceutical industry over the past three years. These drugs, originally developed to treat type 2 diabetes, have proven extraordinarily effective for weight loss, generating demand that has repeatedly outstripped manufacturing capacity. Lilly has invested more than $9 billion in new manufacturing facilities since 2023, including a major expansion at its Lebanon, Indiana plant and a new facility in Research Triangle Park, North Carolina.
Novo Nordisk, Lilly's primary competitor, reported disappointing Q1 results in late April, with Wegovy supply constraints limiting growth. That contrast has sharpened investor focus on Lilly's manufacturing execution as a key competitive differentiator heading into the second half of 2026.
Key Developments
Lilly's Q1 2026 revenue exceeded analyst consensus by a substantial margin, with Mounjaro generating approximately $4.8 billion in quarterly sales -- up 125% from the same period in 2025. Zepbound, the obesity-specific formulation of tirzepatide, contributed roughly $2.3 billion, an 80% year-over-year increase. The company raised its full-year 2026 revenue guidance, signaling confidence that supply constraints that plagued the business in 2024 and early 2025 are now largely resolved.
Chief Executive David Ricks highlighted progress on manufacturing capacity as the primary driver of the earnings beat, noting that Lilly's new facilities are now operating at scale. The company also reported positive early data from clinical trials examining tirzepatide's potential in cardiovascular disease prevention and chronic kidney disease -- indications that could dramatically expand the addressable market beyond diabetes and obesity.
Qualcomm also reported strong results on May 1, with shares surging 11% after the chipmaker disclosed a significant new custom silicon engagement with a major cloud provider, adding to a day of broadly positive earnings sentiment across the S&P 500.
Why Americans Should Care
The Mounjaro and Zepbound sales figures have direct implications for American patients and the healthcare system. More than 38 million Americans live with diabetes, and an estimated 42% of US adults are classified as obese -- making the GLP-1 drug class potentially the most consequential pharmaceutical development in a generation. Access remains a critical issue: Mounjaro carries a list price of approximately $1,000 per month, and insurance coverage varies widely by state and employer. In states like Mississippi, Alabama, and West Virginia -- which have the highest obesity rates in the country -- Medicaid coverage for GLP-1 drugs remains limited, creating a stark disparity between patients who can afford treatment and those who cannot. Congressional Democrats from these states have introduced legislation to expand Medicaid coverage for obesity drugs, a debate that Lilly's earnings results will intensify.
Why It Matters
Eli Lilly's Q1 results represent more than a single company's success -- they signal a structural shift in how the US healthcare system will manage chronic disease over the next decade. The GLP-1 drug class is on track to generate more than $100 billion in annual global revenue by 2030, according to multiple analyst projections, making it the largest drug category in pharmaceutical history. This trajectory has profound second-order effects: reduced demand for bariatric surgery, lower long-term cardiovascular disease costs, and potential reductions in insulin sales -- a market Lilly itself dominates.
Internationally, the US leads GLP-1 adoption by a wide margin, with European regulators moving more cautiously on obesity indications and Asian markets still in early stages of uptake. The pricing gap between the US and other developed markets -- where the same drugs cost a fraction of American list prices -- has become a flashpoint in congressional drug pricing debates, with the Lilly earnings report likely to renew calls for Medicare negotiation authority over GLP-1 drugs. The company's stock has risen more than 60% over the past 18 months, making it one of the most valuable pharmaceutical companies in the world.
What's Next
Lilly is expected to file for FDA approval of tirzepatide in chronic kidney disease by the end of 2026, a decision that could open a market of more than 37 million Americans with the condition. The company's next major clinical readout -- examining cardiovascular outcomes in obese patients without diabetes -- is expected in the third quarter. Novo Nordisk will report its full Q1 results in mid-May, and the comparison with Lilly's performance will set the competitive narrative for the remainder of the year.
Sources: Eli Lilly Investor Relations; Charles Schwab Market Update